The basics of electrospinning
Electrospinning is a continuous low-cost technology to generate dried fibers by utilizing the electrostatic forces on a liquid feed to turn it into ultrafine (normally <10 μm) fiber structures, which can dry instantly at room temperature during process operation.
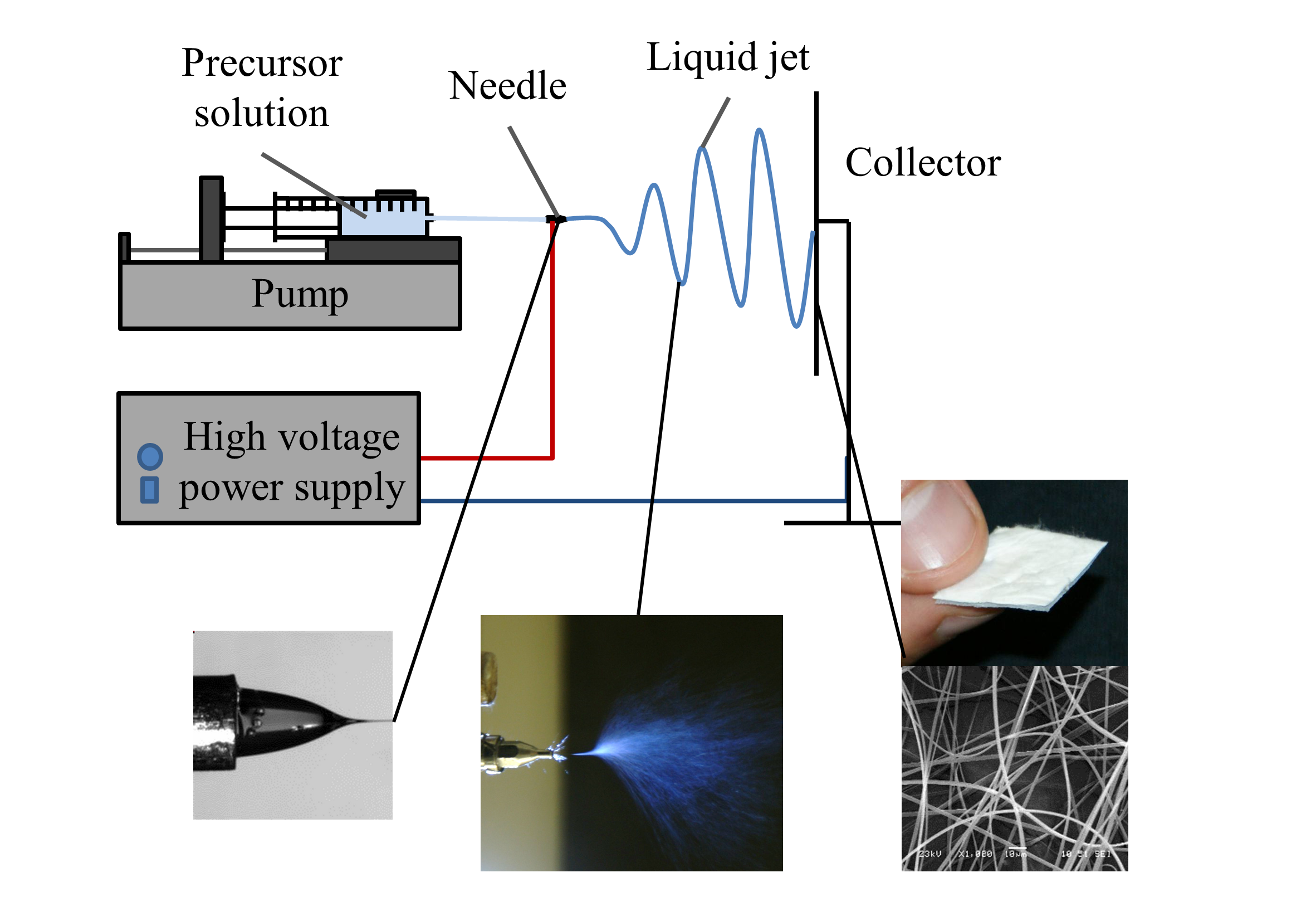
The fiber-forming excipient (polymer or cyclodextrin, lipid, etc.) is dissolved in a solvent and the solution is fed into a single spinneret at a constant, controlled flow rate. High voltage is applied between the spinneret and the grounded collector. When the electrostatic forces overcome the surface tension, a liquid jet breaks out from the liquid surface and stabilizes between the nozzle and the collector as a continuous stream. During the process, the jets get elongated gaining a fiber-like structure. The solvent evaporates instantaneously at room temperature due to the high surface area, as the fibers are often submicron-sized. By using solutions with lower concentrations, it is possible to prepare spherical particles - this process is called electrospraying.

Scaled-up electrospinning
We developed high-speed electrospinning - it utilizes both centrifugal and electrostatic forces for large-scale nano- or microfiber production. By using the flexible pilot-scale GMP-compliant machine, 300-500 g solid product/h production rates can be achieved.

Key features and advantages of high-speed electrospinning include:
- production of nanofibers or particles with ultrafast dissolution;
- increased bioavailability of poorly soluble drugs by creating (nano)amorphous solid dispersions;
- possibility of the drying of sensitive APIs at room temperature;
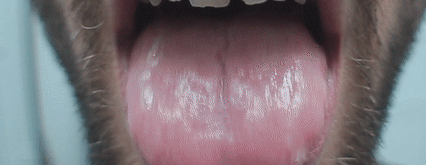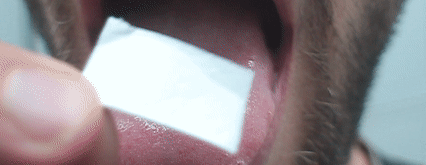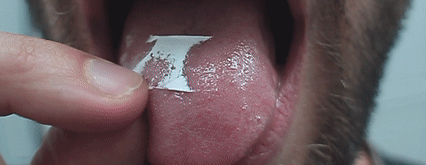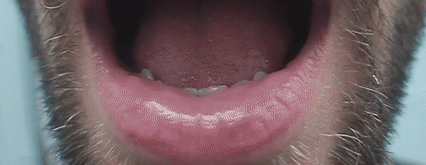
- single-step production of formulations for special drug delivery routes (buccal, intranasal, transdermal, etc.);
- scalability – rapid feasibility studies from 200 mg API to pilot-scale production.
Relevant publications
J. Domján, P. Vass, E. Hirsch, E. Szabó, E. Pantea, S.K. Andersen, T. Vigh, G. Verreck, G. Marosi, Z.K. Nagy, Monoclonal antibody formulation manufactured by high-speed electrospinning, International Journal of Pharmaceutics, 591 (2020) 120042, doi:10.1016/j.ijpharm.2020.120042.
E. Szabó, P. Záhonyi, D. Brecska, D.L. Galata, L.A. Mészáros, L. Madarász, K. Csorba, P. Vass, E. Hirsch, J. Szafraniec-Szczęsny, I. Csontos, A. Farkas, G. Van den Mooter, Z.K. Nagy, G. Marosi, Comparison of amorphous solid dispersions of spironolactone prepared by spray drying and electrospinning: The influence of the preparation method on the dissolution properties, Molecular Pharmaceutics, 18 (2020) 317-327, doi:10.1021/acs.molpharmaceut.0c00965.
P. Vass, E. Pantea, A. Domokos, E. Hirsch, J. Domján, Á. Németh, M. Molnár, Cs. Fehér, S.K. Andersen, T. Vigh, G. Verreck, I. Csontos, G. Marosi, Z.K. Nagy, Electrospun solid formulation of anaerobic gut microbiome bacteria, AAPS PharmSciTech, 21 (2020) 214, doi: 10.1208/s12249-020-01769-y.
K. Kiss, P. Vass, A. Farkas, E. Hirsch, E. Szabó, G. Mező, Z.K. Nagy, G. Marosi, A solid doxycycline HP-β-CD formulation for reconstitution (i.v. bolus) prepared by scaled-up electrospinning, International Journal of Pharmaceutics, 586 (2020) 119539, doi: 10.1016/j.ijpharm.2020.119539.
P. Vass, E. Szabó, A. Domokos, E. Hirsch, D. Galata, B. Farkas, B. Démuth, S.K. Andersen, T. Vigh, G. Verreck, G. Marosi, Z.K. Nagy, Scale‐up of electrospinning technology: Applications in the pharmaceutical industry, WIREs Nanomedicine and Nanobiotechnology, (2019) e1611, doi: 10.1002/wnan.1611.
E. Szabó, B. Démuth, D.L. Galata, P. Vass, E. Hirsch, I. Csontos, G. Marosi, Z.K. Nagy, Continuous formulation approaches of amorphous solid dispersions: Significance of powder flow properties and feeding performance, Pharmaceutics, 11 (2019) 654, doi: 10.3390/pharmaceutics11120654.
P. Vass, Z.K. Nagy, R. Kóczián, C. Fehér, B. Démuth, E. Szabó, S.K. Andersen, T. Vigh, G. Verreck, I. Csontos, G. Marosi, E. Hirsch, Continuous drying of a protein-type drug using scaled-up fiber formation with HP-β-CD matrix resulting in a directly compressible powder for tableting, European Journal of Pharmaceutical Sciences, 141 (2020) 105089, doi: 10.1016/j.ejps.2019.105089.
P. Vass, E. Hirsch, R. Kóczián, B. Démuth, A. Farkas, C. Fehér, E. Szabó, Á. Németh, S.K. Andersen, T. Vigh, G. Verreck, I. Csontos, G. Marosi, Z.K. Nagy, Scaled-up production and tableting of grindable electrospun fibers containing a protein-type drug, Pharmaceutics, 11 (2019) 329, doi: 10.3390/pharmaceutics11070329.
E. Hirsch, P. Vass, B. Demuth, Z. Petho, E. Bitay, S.K. Andersen, T. Vigh, G. Verreck, K. Molnar, Z.K. Nagy, G. Marosi, Electrospinning scale-up and formulation development of PVA nanofibers aiming oral delivery of biopharmaceuticals, Express Polymer Letters, 13 (2019) 590-603, doi: 10.3144/expresspolymlett.2019.50.
P. Vass, B. Démuth, A. Farkas, E. Hirsch, E. Szabó, B. Nagy, S.K. Andersen, T. Vigh, G. Verreck, I. Csontos, G. Marosi, Z.K. Nagy, Continuous alternative to freeze drying: Manufacturing of cyclodextrin-based reconstitution powder from aqueous solution using scaled-up electrospinning, Journal of Controlled Release, 298 (2019) 120-127, doi: 10.1016/j.jconrel.2019.02.019.
P. Vass, B. Démuth, E. Hirsch, B. Nagy, S.K. Andersen, T. Vigh, G. Verreck, I. Csontos, Z.K. Nagy, G. Marosi, Drying technology strategies for colon-targeted oral delivery of biopharmaceuticals, Journal of Controlled Release, 296 (2019) 162-178, doi: 10.1016/j.jconrel.2019.01.023.
